 (Solution 2: NEUTRALS IN METHYLENE CHLORIDE) The acids go into the water layer. The neutrals stay in the methylene chloride layer. Step 3: Take the methylene chloride layer from step (1) and shake this against an aqueous layer with a pH value of 13 (adjusted to that level using a concentrated solution of sodium hydroxide). (Solution 1 – ORGANIC BASES IN METHYLENE CHLORIDE) Step 2: Remove the water layer from step (1), adjust the pH back to a value of 13 using a concentrated solution of sodium hydroxide, shake against methylene chloride, and we now have a solution of the organic bases in methylene chloride. Step 1: Lower the pH of the water using concentrated hydrochloric acid.Įxtract with methylene chloride – the neutrals and acids go into the methylene chloride, the bases stay in the water. Neutrals – Whether the pH is acidic or basic, these will remain neutral under all circumstances.Īt very acidic pH values (say a pH of around 1) – these are fully protonated and neutralĪt basic pH values (say a pH of around 13) – these are fully deprotonated and anionicĪt very acidic pH values (say a pH of around 1) – these are protonated and cationicĪt very basic pH values (say a pH of around 13) – these are not protonated and neutral The key to understanding how to do this separation relates to the effect that pH will have on the different categories of compounds. Neutral substances are more soluble in organic solvents than in water.Ionic substances are more soluble in water than in organic solvents.Devise an extraction procedure that would allow you to perform this bulk separation of the three categories of organic compounds. The desire is to have three solutions at the end, each in methylene chloride, one of which contains only the organic acids, the second contains only the organic bases, and the third contains only the neutrals. The compounds can be grouped into broad categories of organic acids, organic bases and neutral organics. The first is a classic example of an extraction procedure that can be used to separate acids, bases, and neutrals.Īn aqueous sample contains a complex mixture of organic compounds, all of which are at trace concentrations. Next we want to examine some general types of extraction procedures that are commonly used.  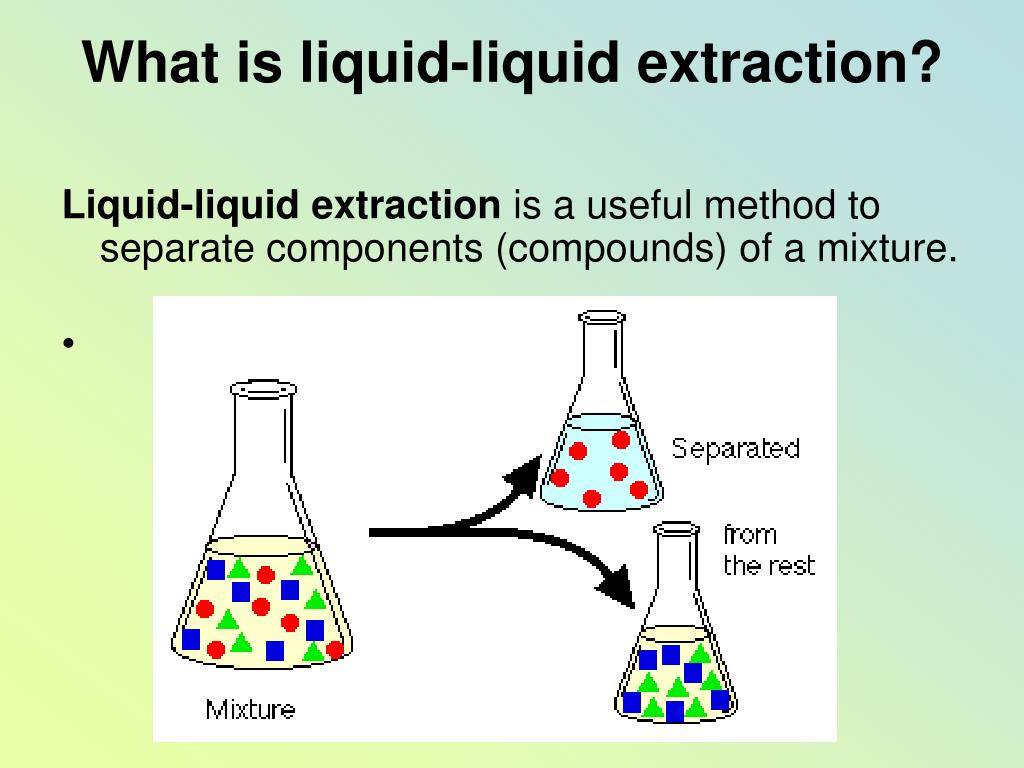
As we will see shortly, this distribution of a solute between two immiscible phases forms the basis of chromatographic separations as well. A fresh portion of the organic phase is then added to remove more of the solute in a second extraction. 
\]įrom experience you have probably had in your organic chemistry lab, you know that the approach that is often used in liquid-liquid extraction is to add some organic phase, shake the mixture, and remove the organic phase. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
